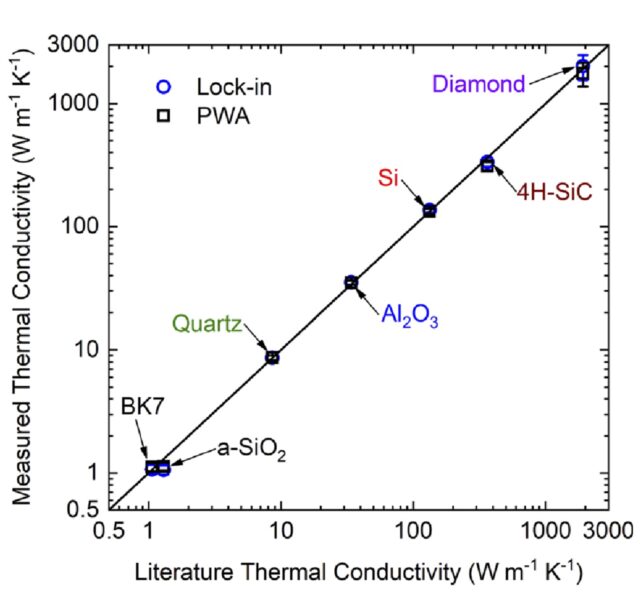
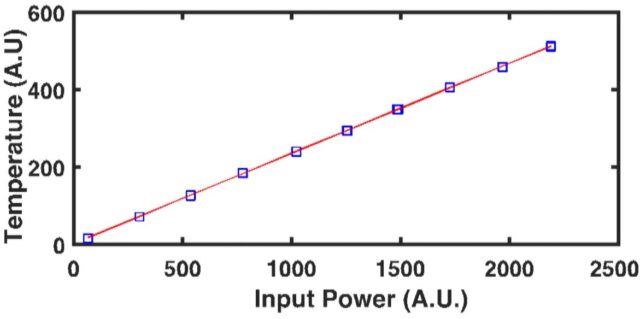
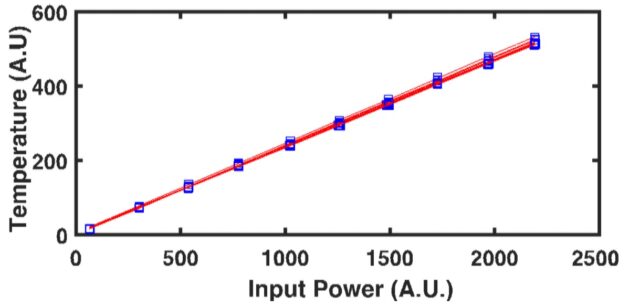
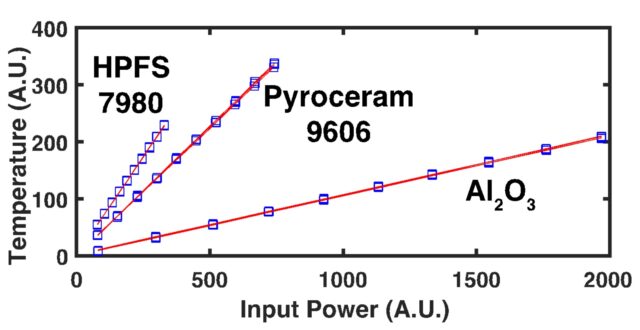
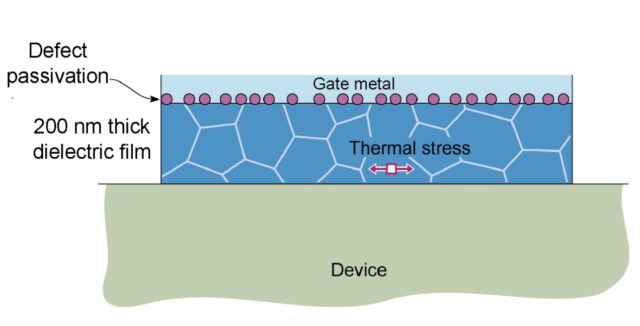
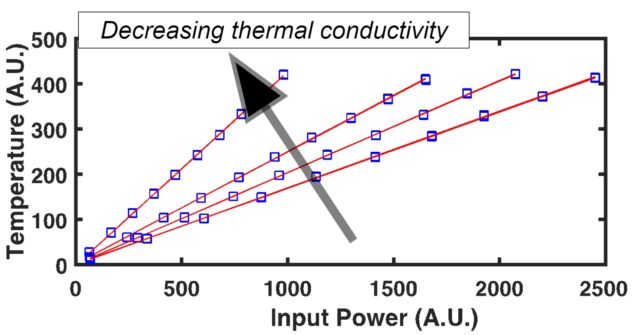
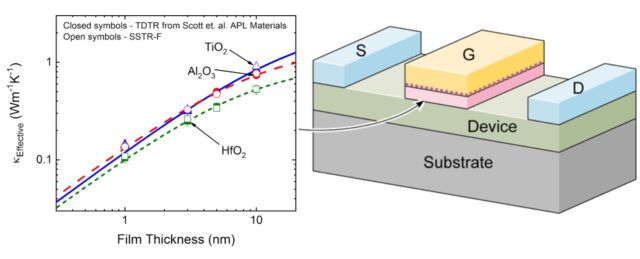
When considering the rate of heat transfer inside a material, we turn to the thermal diffusivity (D or a), which is defined as the ratio of the thermal conductivity to volumetric heat capacity of the material:
D = k/C (m2/s)
Thus, thermal diffusivity provides information about the competition between the ability to conduct heat in a material (k) vs the ability to store that heat (C). It is important to note that a high value of thermal diffusivity does not necessarily mean that heat is better dissipated, but rather that heat is more effectively dissipated than it is stored. Thus, for one to compare materials and assess their ability to dissipate heat, thermal conductivity and thermal resistances should be compared, so that any thermal diffusivity value or measurement must be multiplied by the materials heat capacity. To this end, technically, thermal conductivity measurements provide different information than thermal diffusivity measurements.
As thermal diffusivity is a property that is important during temperature transients, we can then relate the time it takes for heat to travel across some distance, or the time it takes for a material to reach steady state conditions, to the diffusivity, D. The time it takes to travel across a material of given thickness, d, can be approximated by the diffusion time.
tdiffusion> = d2/D
This diffusion time can then be used to approximate the time it takes for a material system to reach steady state. When the time scale of heating is much larger than the diffusion time, the temperature gradients in a material will progress to steady state, and thus the ratio of heating time to tdiffusion gives an indication of whether the material has reached steady state conditions. To this end, we define a non-dimensional number, the Fourier Number (Fo) given by:
Fo = t/tdiffusion = Dt/d2
where steady state conditions are achieved when Fo>>1. Under this condition of Fo>>1, the temperature gradients are not changing in time and the temperature changes across materials can be predicted by the thermal conductivity and thermal resistance.