Thermal conductivity is a form of heat transfer with vital implications for material performance in applications ranging from electronic devices, to manufacturing, to transportation, to even the design of coffee cups.
In this blog, we explain what thermal conductivity is, and what differentiates it from other forms of heat transfer. Accurately measuring thermal conductivity requires an array of different processes, depending on the material being assessed. We conclude with a look at one example of an advanced conductivity measurement technique – Steady State Thermoreflectance (SSTR) – which can measure bulk materials to films as thin as a few nanometers.
Thermal Conductivity Meaning: What is thermal conductivity?
Thermal conductivity (commonly abbreviated using k, λ, or κ) is the ability of a material to transfer (or “conduct”) heat. In short, a material with high conductivity will conduct heat much more efficiently than a material with a low thermal conductivity. Therefore, if you want to prevent your hand from getting too hot when holding a cup of coffee, make sure the coffee cup is made out of a material with a low thermal conductivity!
Thermal conductivity is defined as the quantity that relates the heat flux through the material, Q, to the temperature gradient that is established across the material, dT/dx:
Q=-k dT/dx
This equation, known as “The Fourier Law”, predicts the temperature difference across some thickness of material in steady state conditions (when the temperature is not varying in time). So, if you want to make sure your hand does not get too hot when you are holding your cup of coffee, you can either:
- Make the walls of the coffee cup thicker to make sure your hand is further away from the hot coffee (maybe put a handle on the mug, spacing your hand away from the heat source).
- Make the walls of the coffee cup out of a lower thermal conductivity material (maybe use a coffee sleeve which is made from porous materials with air pockets, making for very low thermal conductivity walls!).
In the International System of Units, thermal conductivity is expressed using W/m*K (Watts per meter-Kelvin). It is one of three ways that heat can transfer through a substance. We concisely explain the difference between each one below.
Three Methods of Heat Transfer: Thermal Conduction Versus Radiation and Convection
-
- Radiation is the transfer of heat via electromagnetic waves. We experience this type of heat whenever we feel the warmth of the sun. Because it relies on electromagnetism, radiation does not require the actual movement of material to transfer heat—which means it can do so in a vacuum (such as outer space).
- Unlike radiation, convection involves the movement of particles in liquid or gas, and often is a primary means of exchanging energy with a surface. “Convective cooling” for example, is a general term used for the process in which an object is cooled based on a gas taking energy away from the surface of the object. If your coffee cup has well insulated walls but you didn’t put a top on it, convective cooling can cause the coffee to cool quickly.
- Conduction describes the transfer of heat without electromagnetic radiation or the bulk movement of the particles themselves. Instead, heat is transferred by the vibration of molecules, atoms, or electrons. Energy moves along a “temperature gradient,” moving from areas of the material with higher temperature (and thus energy) to areas with lower temperature/energy. Thermal conductivity is a quantitative measure of how efficiently this heat is conducted. When steady state is achieved, the temperature gradient does not change with time, and in the simple one-dimensional picture described above, is linear (i.e., the dT/dx term in the Fourier Law)
The rate at which heat is conducted depends on the properties of the material in question. This rate can vary immensely (a highly conductive substance like copper conducts almost 10,000x more heat than air). Measuring thermal conductivity is essential to determining heat transfer rates and temperatures under different conditions.
How is thermal conductivity measured?
The different methods used to test conductivity for various different materials can be broadly grouped into two categories:
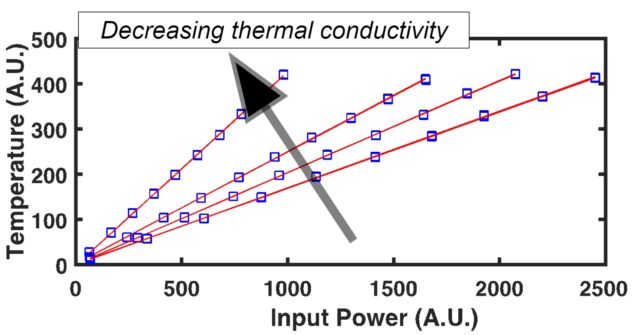
- Steady State: this method captures a measurement once the temperature gradient in a sample is constant in time. After heat is applied and a steady state temperature gradient is achieved after some time, the temperature gradient is measured and directly related to the thermal conductivity.
- Transient: measurements are taken as a function of time during or after a heating process, when the temperature at a point in space of a material is not constant, and hence, the temperature gradients are changing in time. These measurements can be taken using different transient heater and sensor designs such as a “hot wire,” “plane source,” or transient laser-based measurements.
For precision engineered materials requiring highly accurate, reproducible thermal conductivity data, finding the appropriate measurement methodology can be a real challenge. Ultra-thin films are a great example.
Measuring Thin Film Thermal Conductivity
Most traditional thermal conductivity measurement techniques use heat sources or sensors (thermometers) that are often orders of magnitude larger than the thickness of the films that are ubiquitous in modern micro- and nanotechnologies. This means that the volume of material being measured using these techniques is much greater than the film of interest and, thus, the temperatures and temperature gradients being measured are dominated by everything but the thin film. Consequently, these techniques are not suitable to accurately measure the thermal conductivity of thin film materials.
As a result, only selected techniques with heaters and/or sensors that are designed with temporal and/or spatial resolutions that can capture the thin film temperature gradients are suitable for measuring thin film thermal conductivity. This is typically achieved using advanced metal patterning or focused laser systems.
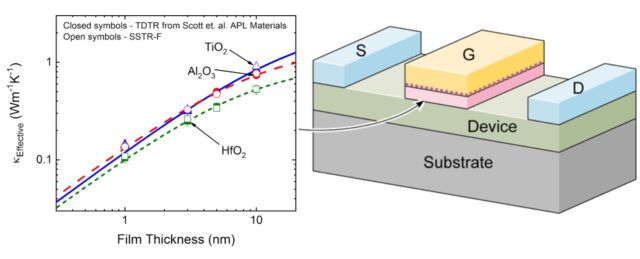
Steady-State Thermoreflectance in Fiber Optics (SSTR-F) provides one example of an advanced thin-film measurement solution. SSTR offers the unprecedented, nanometer-precise spatial resolution needed to effectively measure thermal conductivity in thin films. It can also be leveraged for bulk wafer and substrate testing needs.
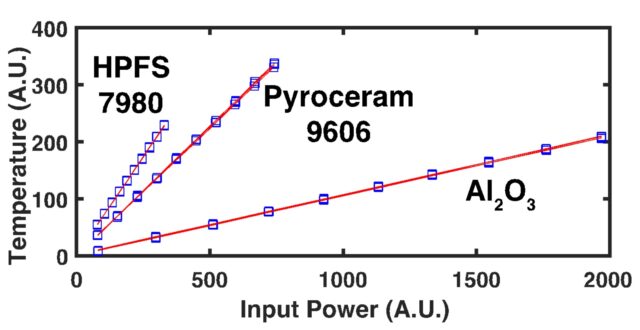
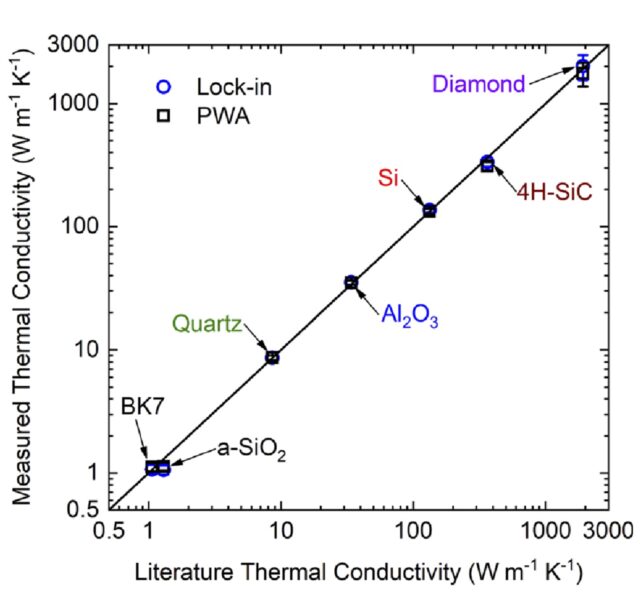
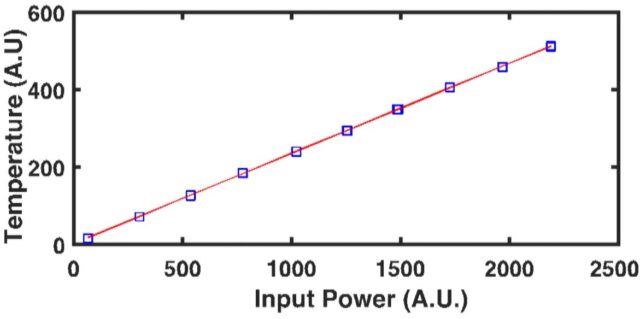
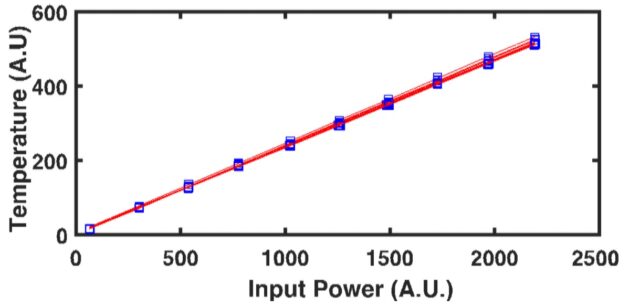
Through the use of a patented1 fiber-optic design, SSTR-F uses a laser to create a heating event on the surface of a sample, which results in a steady-state temperature gradient that is established under the sample surface. A secondary probe laser then measures the temperature on the surface of the sample resulting from different steady state temperature gradients that are established from different pump powers. Returning to Fourier’s Law, the heating event provides the “Q,” and the probe laser measures differences in dT/dx based on different “Q’s” in steady state. With these two properties, SSTR-F measures thermal conductivity. The key in this thin film metrology is that the laser spot size can be tuned to be as small as a few microns 1/e^2 diameter, resulting in the ability to measure the thermal conductivity of even extremely thin films.
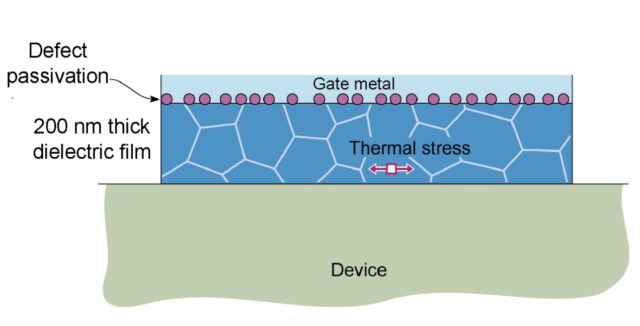
SSTR-F can rapidly detect variation in thermal conductivity that occurs due to defects, stresses, or compositional inconsistencies. This approach is able to achieve:
- Accuracy +/- 10%
- Repeatability +/- 0.5%
- Reproducibility +/- 1%
1: B. M. Foley, J. T. Gaskins and P. E. Hopkins, “Fiber-optic based thermal reflectance material property measurement system and related methods,” United States Patent Number US, 10,928,317 B2 (2021).
Learning More About Measuring Thin Film Thermal Conductivity
If you’re interested in seeing an example of the sort of results that SSTR-F can produce in regards to thermal conductivity testing, please see our validated sample results here.
Or, if you are interested in learning more about SSTR-F, the Laser Thermal team is happy to chat about how we can help solve your measurement challenges.